The Jacobs Lab
Research Areas
We currently have three areas of focus. 1) Mechanisms of epilepsy and hyperexcitability in developmental cortical malformations. 2) Physiological abnormalities after traumatic brain injury and whether they are resolved with specific treatments. 3) Posttraumatic epilepsy after repeated trauma or with pre-existing conditions of abnormal development.
Epilepsy associated with developmental cortical malformation
Using a neonatal transcranial freeze lesion, we have replicated the histopathology and hyperexcitability of 4-layered polymicrogryia associated with epilepsy. We have documented a number of intrinsic cellular, synaptic and network abnormalities. We are currently using a combination of optogenetics and electrophysiology to test our novel hypothesis that increased function of an inhibitory interneuron subtype promotes the epileptiform activity. In addition, we are testing several potential treatments in this highly translational work.

Microgyrus and sulcus induced in rodent neocortex.
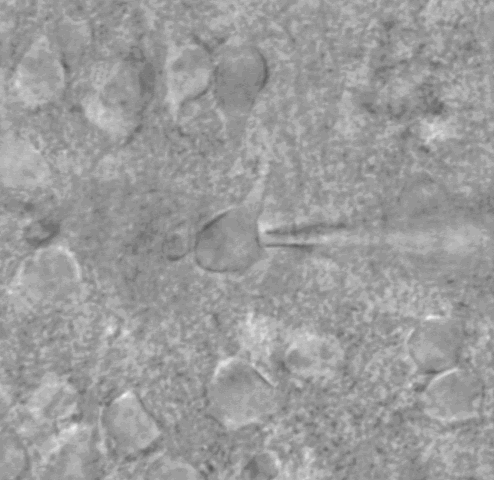
Whole cell patch clamp under DIC optics


Biocytin-filled pyramidal neuron.
Action potential firing patterns typical of two types of inhibitory interneurons: FS = fast-spiking, parvalbumin-containing, and LTS = low threshold-spiking, typically containing somatostatin.

Top: Epileptiform activity recorded as local field potentials in ex vivo slices. Dots show timing of electrical stimulation. Purple arrows show abnormal (epileptiform) events. Bottom: Optically activated IPSCs recorded from a layer V pyramidal neuron in neocortex with Channelrhodopsin in Somatostatin inhibitory interneurons. Right: cartoon of hypothesized cellular network.
Mild Traumatic Brain Injury - physiological consequences
Currently funded by the National Institute of Neurological Disorders and Stroke (NIH - NINDS)
Using a central fluid percussion injury, we have replicated the key feature of mild brain injury, that of diffuse axonal injury (DAI). We have found that despite decades of focus specifically on these axotomized neurons that the structurally intact neurons also function abnormally in the injured brain. Within two days of the injury, there are abnormalities in the action potentials, the frequencies of firing and synaptic activity. Excitatory input to layer V pyramidal neurons of the neocortex is increased, likely contributing to the hyperexcitability of the local network. Our data show that some of these abnormalities are long-lasting, perhaps underlying the chronic symptoms experienced by patients. We are currently utilizing a combination of whole cell patch clamp, optogenetics, field potential recordings, immunohistochemistry and Western blots to identify the mechanisms of these abnormalities. We are also examining whether treatments that show promise in alleviating symptoms change these cellular and network anomalies.

Using YFP-H strain of trangenic mice allows for visualization of neuronal morphology in the living slice. YFP fills a subset of layer V pyramidal neurons. Most have a regular-spiking adapting firing pattern, but some are also intrinsically-bursting (IB) neurons. Not only the soma, but the status of the axon can be seen prior to choosing a neuron for recording. In A. the arrow points to a swelling along the axon, that is a site of disconnection with the distal portion of the axon. In B, a different neuron, having multiple swellings but ultimate disconnection is shown. For some neurons the axotomy occurred further from the soma, as shown in C. The recorded neurons were filled with biocytin. D shows the biocytin reaction for an axotomized neuron. In E the same neuron as in D, but showing the YFP, where a second (non-recorded) cell is shown at the arrowhead. The second neuron has YFP but no biocytin (not observed in D). The overlap is shown in F. In Hand I are examples of intact neurons, where the axon could be followed to the subcortical white matter.
Posttraumatic Epilepsy induced by mild traumatic brain injury
Grant currently being reviewed
There are currently no in vivo animal models of epilepsy induced by mild traumatic brain injury. Single mild injuries do not produce seizures in otherwise normal animals. Although the likelihood of seizures after severe injury is much higher than that after mild injury, there are many thousands more mild injuries occurring annually, so that the actual number of patients that develop epilepsy after mild traumatic brain injury is much higher than that after severe injury. Thus a model of epilepsy after mild injury is needed. We hypothesize that the history of brain injury and neurological condition of the brain at the time of injury affects the likelihood of developing epilepsy. We are examining the potential of repeated mild traumatic brain injury for the subsequent development of epilepsy.